Rapamycin anti-aging benefits are no longer confined to biohacker forums or speculative longevity podcasts. They are now the subject of rigorous peer-reviewed clinical trials, landmark human studies, and a rapidly expanding body of evidence published in the world's most respected scientific journals — including Science Translational Medicine, The Lancet Healthy Longevity, and a groundbreaking 2026 study from the University of Oxford.
The rapamycin anti-aging benefits that researchers are documenting in 2025 and 2026 go far beyond anything imagined a decade ago. By precisely inhibiting the mTOR pathway — the master molecular switch governing cellular growth, repair, and the critical self-cleaning process called autophagy (from the Greek for "self-eating") — low-dose intermittent rapamycin is achieving something once considered biologically impossible: a measurable, clinically documented reversal of immunological aging in living humans.
This is not about slowing immune decline. This is about turning the biological clock backward — and the science behind it is now extraordinary.
2026 Research Update: A landmark University of Oxford study found that low-dose rapamycin helps T-cells (the white blood cells that coordinate immune responses) resist DNA damage — a previously undiscovered rejuvenation mechanism that adds a powerful new dimension to the rapamycin anti-aging benefits profile.
Buy Rapamycin online
What Is the mTOR Pathway and Why Does It Drive Aging?
Every discussion of rapamycin anti-aging benefits begins with one protein: mTOR — mechanistic Target of Rapamycin. This protein kinase (an enzyme that activates other proteins by adding phosphate groups to them) functions as your body's master metabolic sensor and decision-maker.
At every moment, mTOR reads your nutrient availability, energy levels, growth factor signals, and stress indicators, then makes a fundamental decision for every cell: grow and build, or conserve and repair. In youth, this balance is elegantly adaptive. In aging, it fails catastrophically.
The Chronic Overactivation Problem
As we grow older, mTOR becomes chronically and inappropriately overactive — a biological accelerator that cannot be released. The consequences cascade through every system in the body:
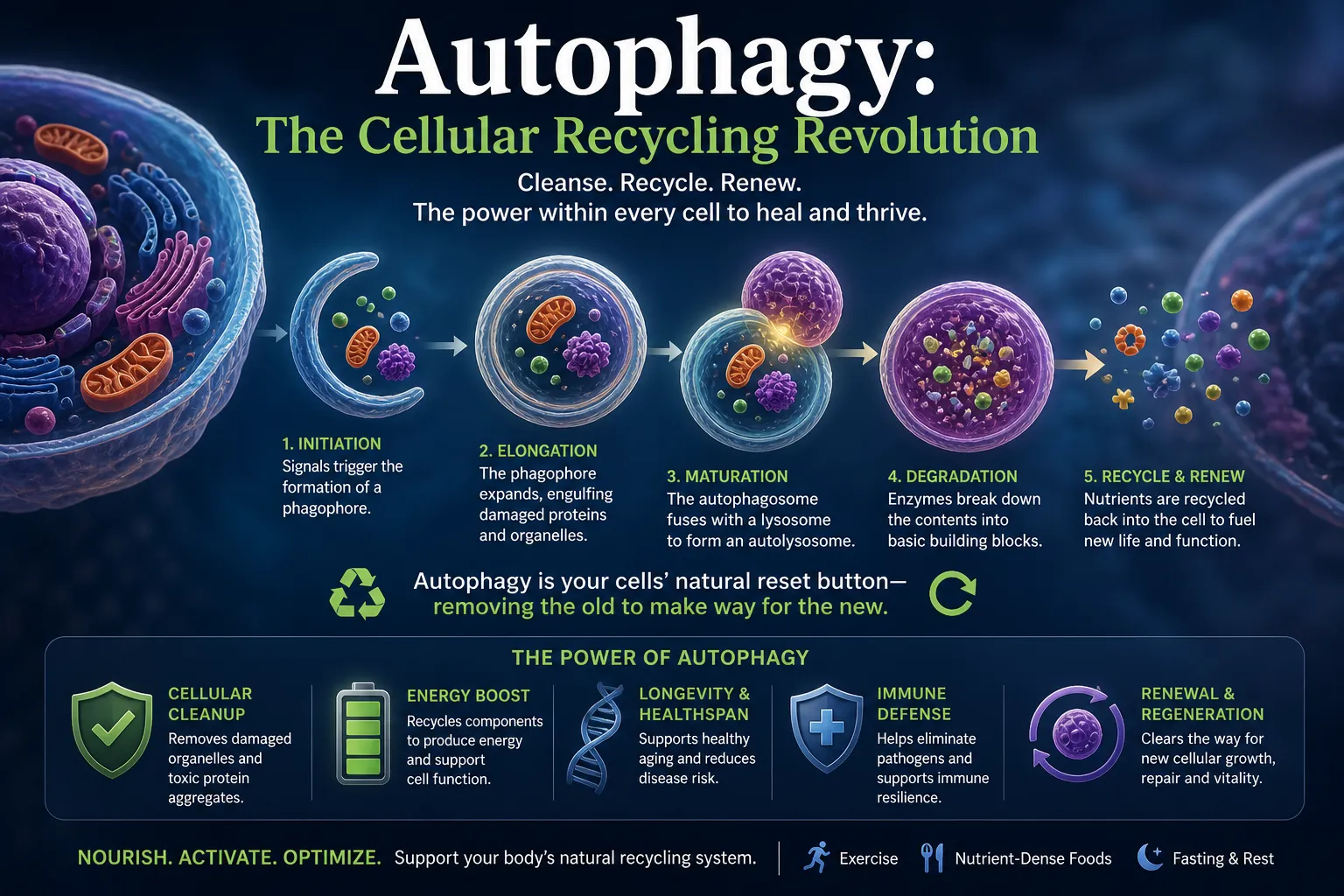
- Autophagy is suppressed. The cell's internal cleaning system — which breaks down damaged proteins, worn-out organelles (the tiny functional units within cells), and molecular debris — is shut off. Cellular "garbage" accumulates, degrading function across every tissue. The Nobel Prize in Medicine was awarded in 2016 for discovering autophagy's mechanisms, underlining its critical importance.
- Senescent "zombie cells" multiply. Cells that have stopped dividing but refuse to die accumulate throughout the body, continuously leaking toxic inflammatory signals — the SASP (senescence-associated secretory phenotype) — that accelerate aging in surrounding tissue.
- T-cells deteriorate. T-cells — the white blood cells that coordinate and execute immune responses — become exhausted, dysfunctional, and unable to recognize new threats.
- Inflammaging entrenches. The chronic, low-grade systemic inflammation that researchers call "inflammaging" takes hold silently, fueling cardiovascular disease, Alzheimer's disease, diabetes, and cancer over decades.
- Immune stem cells deplete. The populations responsible for replenishing immune cells gradually lose their regenerative capacity.
Rapamycin directly and specifically inhibits mTOR. That singular action — precisely modulated by dose and frequency — is the mechanism from which all rapamycin anti-aging benefits flow.
Buy Rapamycin online
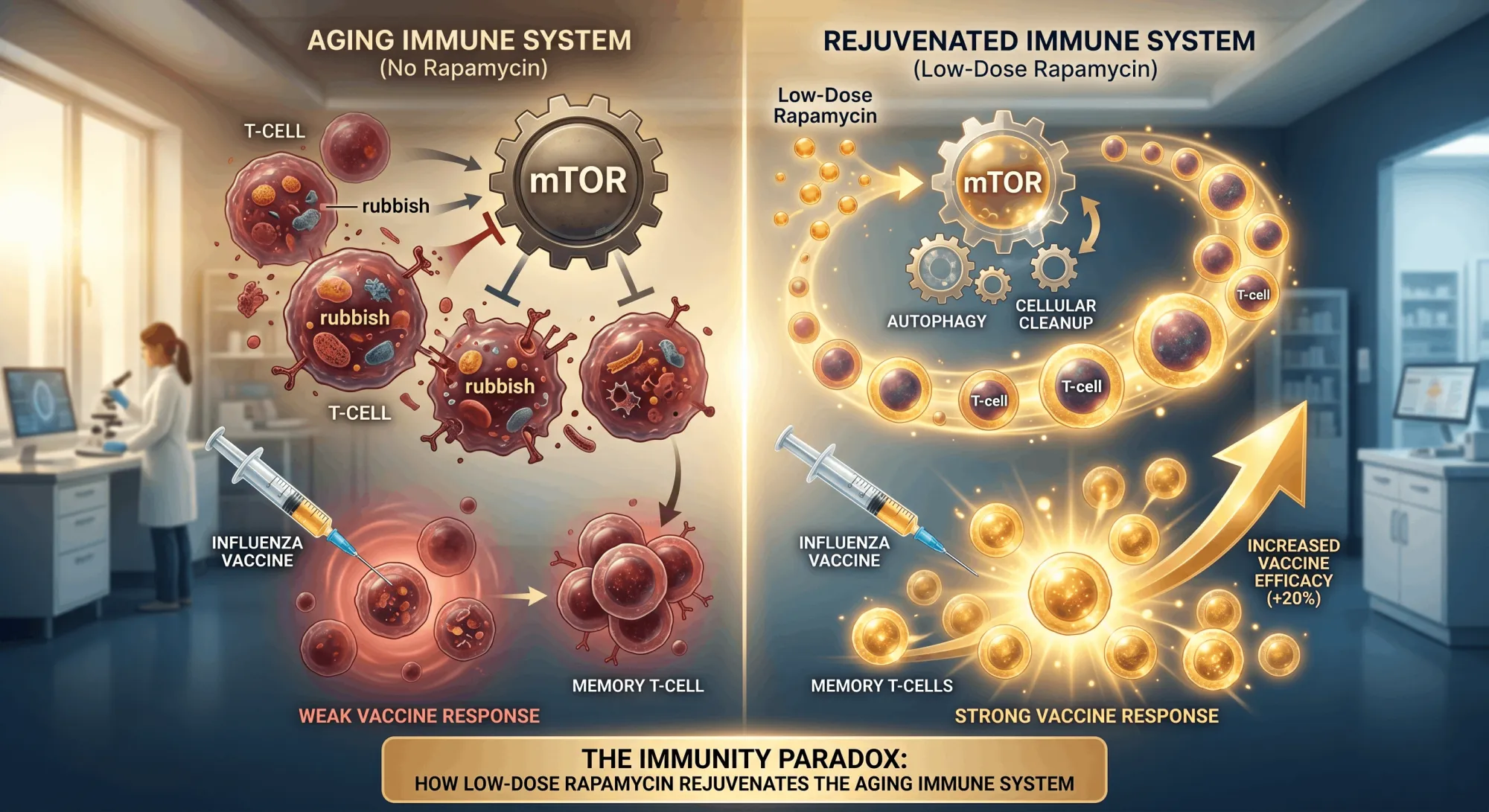
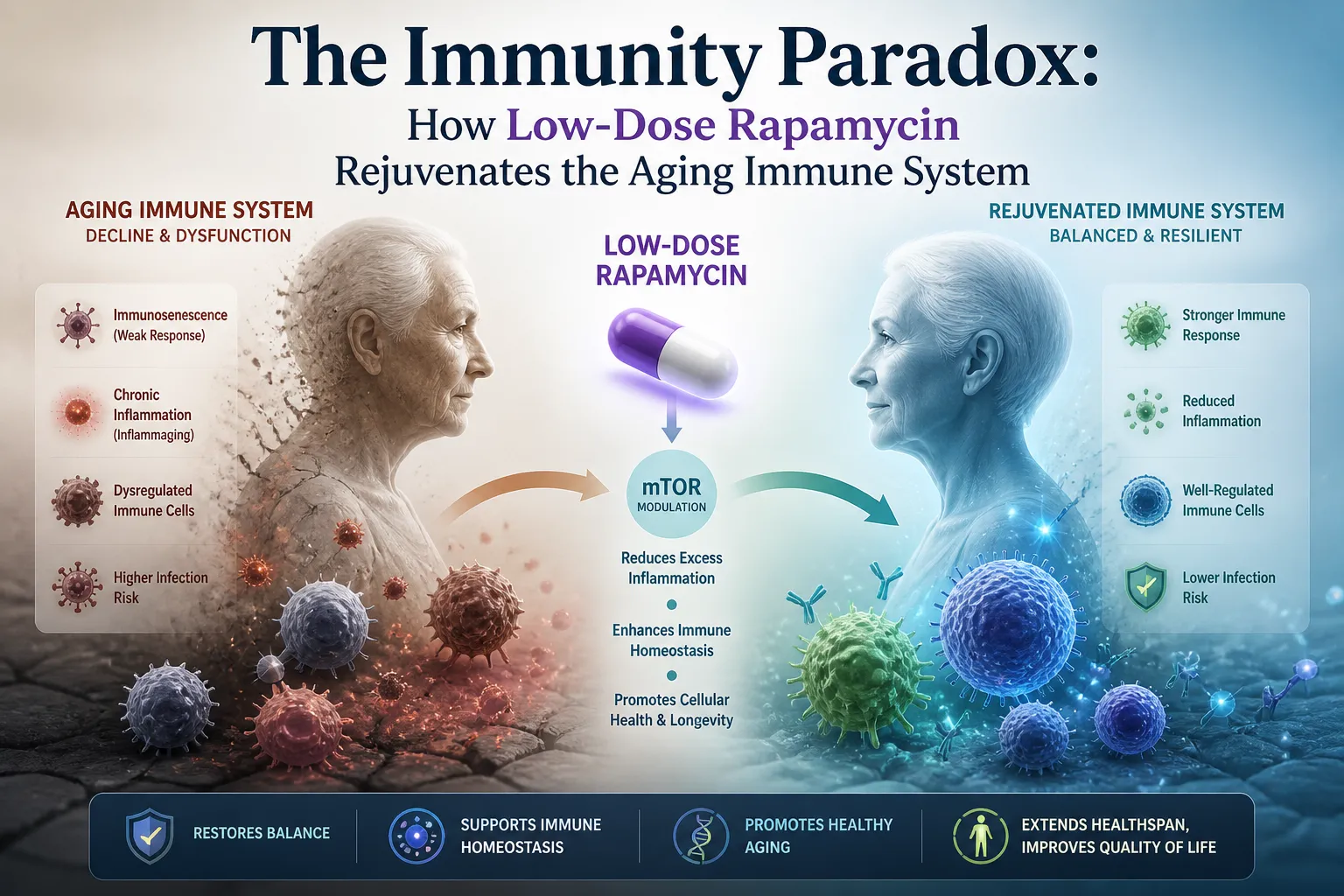
The Immunity Paradox: How a Suppressor Becomes a Rejuvenator
For 30 years, rapamycin was known exclusively as a powerful immunosuppressant — the drug that kept organ transplant recipients alive by preventing rejection. The idea that it could rejuvenate the immune system struck most physicians as paradoxical to the point of absurdity.
The paradox resolves entirely when you understand dose.
At high, continuous doses (2–5 mg daily, as transplant patients take), rapamycin broadly suppresses mTOR across all immune cells, blunting immune activity. At low, intermittent doses — typically 1–6 mg once per week — something fundamentally different occurs: a precise recalibration of mTOR signaling that restarts autophagy, clears immune cell damage, and resets the immune system to a more youthful, vigorous state.
The same drug. Completely opposite biological outcomes. Entirely determined by dose and frequency.
The Complete Spectrum of Rapamycin Anti-Aging Benefits
Benefit 1: Autophagy Restoration — The Nobel Prize-Winning Mechanism
The most foundational rapamycin anti-aging benefit is the restoration of autophagy — the Nobel Prize-winning cellular cleaning process that chronic mTOR overactivation progressively shuts down with age.
By gently inhibiting mTOR, low-dose rapamycin releases the suppressive brake on autophagy. In immune cells specifically, this is transformative: damaged proteins and worn-out organelles are cleared, and cells that were cluttered and sluggish begin functioning with the efficiency of significantly younger cells. This single mechanism underpins virtually every other benefit in this profile.
Benefit 2: T-Cell Rejuvenation — Including the 2026 Oxford Discovery
T-cells are the command-and-control soldiers of the adaptive immune system — the branch of immunity that learns, remembers, and mounts targeted responses to specific threats. Age progressively devastates T-cell populations in multiple ways simultaneously:
- The thymus (the organ where T-cells mature) shrinks dramatically each decade, producing fewer and fewer new T-cells
- The pool of naive T-cells (fresh, untrained cells capable of responding to entirely new pathogens) becomes severely depleted
- T-cells accumulate DNA damage — the focus of the landmark 2026 Oxford University study — that impairs their ability to divide and mount effective responses
- Senescent T-cells actively broadcast SASP inflammatory signals that drive systemic inflammaging
Rapamycin anti-aging benefits for T-cell rejuvenation operate through multiple simultaneous mechanisms. Restored autophagy clears the accumulated molecular damage driving T-cell senescence. The 2026 Oxford discovery adds a newly identified layer: low-dose rapamycin measurably enhances T-cell resilience against DNA damage, providing protection that helps preserve T-cell functional capacity far longer. Clinical studies document improved T-cell diversity indices — a direct measure of how many different threats the immune system can recognize and respond to.
Benefit 3: B-Cell Enhancement and Superior Antibody Production
B-cells produce the antibodies that neutralize viruses, bacteria, and other pathogens. Aging comprehensively impairs B-cell function: antibody diversity decreases, the germinal center reaction (the critical process by which B-cells rapidly evolve to produce increasingly potent antibodies) weakens, and the binding precision of produced antibodies — their ability to target pathogens accurately — declines.
Low-dose rapamycin restores the germinal center reaction, enabling B-cells to undergo the rapid evolutionary optimization that generates high-quality, precisely targeted antibodies. This is the direct mechanism behind one of the most practically significant rapamycin anti-aging benefits: dramatically improved vaccine responses in older adults, proven in rigorous human clinical trials.
Benefit 4: Inflammaging Reversal
Inflammaging — a term coined by immunologist Claudio Franceschi — describes the chronic, low-grade systemic inflammation that develops progressively with aging and is now recognized as a central molecular driver of virtually every major age-related disease.
It is not the beneficial, self-limiting inflammation of an infection or injury. It is a persistent background fire that smolders for decades — fueled primarily by senescent zombie cells leaking SASP inflammatory cytokines (protein signaling molecules such as interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-alpha), and C-reactive protein (CRP)) into the bloodstream.
Low-dose rapamycin reverses inflammaging through two complementary pathways: restored autophagy clears the damaged molecular components that trigger SASP; and recalibrated mTOR signaling directly reduces cytokine overproduction in immune cells. Multiple clinical studies document measurable reductions in circulating IL-6 and CRP — the two most clinically reliable biomarkers of systemic inflammation and predictors of long-term disease risk.
Benefit 5: Cancer Immunosurveillance Restoration
Every day, the human body produces cells carrying DNA mutations that could develop into cancer. In immunologically young individuals, T-cells and natural killer cells (innate immune cells that specialize in detecting and destroying abnormal cells) identify and eliminate the vast majority of these aberrant cells before they can establish a tumor.
As immunosenescence — the age-related deterioration of the immune system — progressively impairs T-cell and natural killer cell function, this surveillance capacity declines. This is a primary biological reason why cancer incidence rises so sharply with age. By rejuvenating T-cell and natural killer cell function, low-dose rapamycin may help restore this critical anti-cancer patrol. Multiple animal longevity studies have documented significantly reduced cancer incidence in rapamycin-treated subjects, and human clinical investigation is actively underway.
Buy Rapamycin online
The Clinical Evidence: Landmark Human Studies
The Novartis Everolimus Trial (2014) — Proof of Concept
Published in Science Translational Medicine, this landmark trial was the first human proof that mTOR inhibition can genuinely restore immune function in aging adults. Healthy elderly volunteers received low-dose everolimus — a rapamycin analogue with identical mTOR-inhibiting properties — for six weeks, then received the influenza vaccine.
Result: up to 20% stronger antibody response compared to the placebo group. The individuals with the weakest baseline immune function showed the greatest improvements — proving for the first time in humans that the Immunity Paradox of rapamycin is clinically real and practically meaningful.
The Joan Mannick Clinical Program — Building the Evidence Base
Subsequent trials led by Dr. Joan Mannick and her team built systematically on the Novartis findings across multiple studies. They documented consistent improvements in immune gene expression profiles, clinically meaningful reductions in upper respiratory infection rates, enhanced immunological resilience markers, and improved responses to multiple vaccine antigens — all pointing unambiguously toward immune rejuvenation, not suppression, at low intermittent doses.
The 2026 Oxford University DNA Protection Study — A New Frontier
The most recent major advance in rapamycin anti-aging benefits research identified a previously undescribed mechanism: enhanced T-cell resilience against DNA damage accumulation. This discovery explains immune improvements that prior autophagy-focused research could not fully account for, adding a significant new scientific dimension to understanding how rapamycin achieves its rejuvenating effects.
The Lancet Healthy Longevity Systematic Review — Population Validation
A comprehensive review of 19 human studies confirmed that rapamycin and its analogues demonstrably improve immune, cardiovascular, and skin aging parameters in human subjects. Critically, the review found no serious adverse events attributable to rapamycin in healthy individuals at the doses studied across all 19 trials — a foundational safety finding for the anti-aging use case.
Animal Longevity Studies — Remarkable Consistency
Rapamycin administered to middle-aged mice has extended median lifespan by 9–14% — among the largest pharmacological lifespan extensions ever documented in any mammalian model. These animals demonstrated measurably better immune function, cognitive performance, and cancer resistance well into advanced old age. The finding has been independently replicated across multiple laboratories, mouse strains, and dosing protocols — a level of reproducibility rare in longevity research.

Buy Rapamycin online
Precision Dosing: The Science Behind the Immunity Paradox
The rapamycin anti-aging benefits described throughout this guide are entirely and critically dose-dependent. Understanding this is essential — the difference between immune suppression and immune rejuvenation is not a different drug, but a different dose and schedule.
Transplant medicine uses 2–5 mg daily — sustained, pervasive mTOR suppression producing broad immunosuppression. Longevity medicine uses 1–6 mg once per week, often with scheduled breaks every few months — an intermittent pulse that triggers autophagy and recalibrates immune function without the sustained suppression of daily dosing.
The biological logic behind the weekly pulse is elegant: it pharmacologically mimics the intermittent periods of caloric restriction and nutrient scarcity that evolutionary biology designed us to experience — periods when mTOR naturally quiets, autophagy naturally increases, and cellular maintenance takes priority. In the modern world of perpetual food abundance, these reset windows rarely occur naturally. Low-dose rapamycin recreates them on a controlled, predictable schedule.
Medical Disclaimer: All rapamycin use for anti-aging or immune optimization requires qualified medical supervision. Dosing is highly individualized based on age, health status, genetic factors, body composition, and concurrent medications. This article is educational and does not constitute medical advice. Never self-medicate with rapamycin or any prescription drug.
Rapamycin Anti-Aging Benefits Across the Hallmarks of Aging
Geroscientists have formalized the core biological processes driving aging into what they call the Hallmarks of Aging. The extraordinary scope of rapamycin anti-aging benefits becomes fully apparent when mapped against this framework — because rapamycin simultaneously addresses more hallmarks than any other single pharmacological intervention currently known:
- Disabled macroautophagy — directly and powerfully reversed through mTOR inhibition
- Cellular senescence — reduced through autophagy-mediated clearance of SASP-producing zombie cells
- Deregulated nutrient sensing — corrected at its source by recalibrating mTOR itself
- Inflammaging — reduced through lower cytokine production and SASP suppression
- Stem cell exhaustion — partially addressed through improved cellular maintenance programs
- Mitochondrial dysfunction — improved via mitophagy (the specific autophagy of damaged mitochondria — the energy-producing organelles within cells)
- Genomic instability — addressed through the 2026 Oxford T-cell DNA damage protection discovery
Most longevity interventions — pharmaceutical or lifestyle — target one or two of these hallmarks. Rapamycin, through its central modulation of the mTOR pathway, simultaneously influences seven or more. This multi-hallmark targeting is what places it in a category of its own in the science of aging.

Get Rapamycin now
Frequently Asked Questions About Rapamycin Anti-Aging Benefits
Q1: What are the main rapamycin anti-aging benefits for the immune system?
The primary rapamycin anti-aging benefits include: autophagy restoration in immune cells; T-cell rejuvenation through molecular damage clearance and new DNA damage resistance (per the 2026 Oxford study); enhanced B-cell antibody production; dramatically improved vaccine responses in older adults (up to 20% in human trials); measurable reversal of inflammaging; and restoration of cancer immunosurveillance. All benefits stem from one mechanism: precise mTOR inhibition at low, intermittent doses.
Q2: How does rapamycin rejuvenate immunity when it is classified as an immunosuppressant?
This is the Immunity Paradox. High daily doses (transplant medicine) suppress mTOR broadly, blunting immune responses. Low weekly doses recalibrate mTOR signaling — restarting autophagy, clearing immune cell damage, reducing inflammaging, and restoring T-cell and B-cell function. The immune system ends up stronger and more youthful. The determining factors are dose and frequency, not the drug itself.
Q3: What is autophagy and why is it central to rapamycin anti-aging benefits?
Autophagy is the cellular recycling program — identified by 2016 Nobel Prize laureate Yoshinori Ohsumi — through which cells identify, engulf, and break down damaged proteins, dysfunctional organelles, and molecular debris. Chronic mTOR overactivation with aging suppresses autophagy progressively, causing accumulation of cellular dysfunction. Restoring autophagy via rapamycin allows immune cells to clean themselves and function as though they are significantly younger.
Q4: What does the clinical evidence show about rapamycin and vaccine responses?
The evidence is compelling and human-proven. The 2014 Novartis everolimus trial documented up to a 20% improvement in influenza vaccine antibody titers in elderly adults receiving low-dose mTOR inhibition. Dr. Joan Mannick's subsequent trials confirmed improved immune gene expression and reduced respiratory infection rates. Research into shingles, RSV, pneumococcal, and COVID-19 booster vaccine responses is actively ongoing.
Q5: What is the right low dose of rapamycin for anti-aging benefits?
Current longevity protocols typically study 1–6 mg once per week with periodic breaks — fundamentally different from the 2–5 mg daily of transplant medicine. There is no universally correct dose. Optimal dosing depends on age, health status, body composition, genetics, and concurrent medications. All dosing must be individually determined by a qualified physician with regular blood monitoring.
Q6: What are the side effects of low-dose rapamycin?
At longevity doses, rapamycin is generally well tolerated. The most commonly reported effects are mild mouth sores (stomatitis — ulcerations of the oral mucosa), transient changes in lipid profiles, and minor insulin signaling adjustments warranting glucose monitoring in at-risk individuals. The Lancet systematic review found no serious adverse events attributable to rapamycin in healthy individuals across 19 studies. Side effects are significantly more common at transplant-level daily dosing than at weekly longevity dosing.
Q7: What is inflammaging and how do rapamycin anti-aging benefits address it?
Inflammaging is the chronic, low-grade systemic inflammation that develops progressively with aging, driven by senescent zombie cells leaking SASP inflammatory signals and by dysregulated immune cytokine production. It is now recognized as a central driver of cardiovascular disease, Alzheimer's disease, type 2 diabetes, and cancer. Rapamycin reduces it through two pathways: autophagy restoration clears SASP-triggering cellular debris; and recalibrated mTOR signaling reduces overproduction of inflammatory cytokines such as IL-6 and CRP.
Q8: Who benefits most from rapamycin anti-aging immune effects?
Current evidence shows the strongest documented benefits in adults over 60 experiencing measurable immunosenescence — characterized by reduced vaccine responses, increased respiratory infection susceptibility, elevated inflammatory biomarkers (IL-6, CRP), and reduced T-cell diversity. Research is also actively investigating whether beginning protocols in the mid-50s, before significant immunosenescence develops, offers additional preventive advantages.
Q9: Can rapamycin anti-aging benefits be enhanced with other longevity interventions?
Yes, and combination strategies are among the most actively researched areas in longevity medicine. Documented synergies are being studied with senolytics (drugs such as dasatinib and quercetin that eliminate senescent zombie cells), NAD+ precursors such as NMN and NR (nicotinamide mononucleotide and nicotinamide riboside — precursors to NAD+, the critical cellular energy cofactor), and metformin (which activates the complementary AMPK cellular energy sensing pathway, also promoting autophagy).
Q10: Is rapamycin approved for anti-aging use and how safe is it long term?
Rapamycin (sirolimus) carries FDA approval for organ transplant immunosuppression, lymphangioleiomyomatosis, and certain cancers. Its use for longevity and immune optimization is currently off-label — prescribed by physicians based on scientific evidence beyond formal indications. The Lancet review found no serious adverse events in healthy individuals at longevity doses across 19 human studies. Long-term safety data in healthy adults is still accumulating, underscoring why qualified physician oversight is not optional but essential.
Conclusion: Rapamycin Anti-Aging Benefits and the Future of Immune Medicine
The rapamycin anti-aging benefits documented across more than two decades of research — and accelerating powerfully through 2025 and 2026 — represent one of the most profound paradigm shifts in the history of medicine. For the first time, a pharmacological intervention with a rigorously understood molecular mechanism, a growing human clinical evidence base, and a demonstrated multi-hallmark impact on aging biology offers the genuine scientific prospect of reversing — not merely slowing — key aspects of immune aging.
The immunosuppressant becomes an immune rejuvenator. The transplant drug becomes longevity medicine's most powerful instrument. Restored autophagy clears decades of accumulated cellular debris. Rejuvenated T-cells regain their vigilance and their capacity to protect. Inflammaging — the silent fire of aging — is measurably extinguished. Cancer immunosurveillance is restored. And across all of it, the biological evidence points toward an immune system behaving as if it has reclaimed years it had lost.
As precision dosing protocols are refined, human clinical trials expand, and the field of geroscience matures, the rapamycin anti-aging benefits documented today stand as a defining achievement of proactive longevity medicine — and a compelling preview of what science-driven immune rejuvenation can achieve in the decades ahead.







Sign in to leave a comment.