Membrane proteins are crucial components of biological systems, playing vital roles in cellular communication, transport, and signal transduction. They are key targets for a vast number of drugs. However, determining the high-resolution 3D structures of membrane proteins has historically been a significant challenge for structural biologists. Their hydrophobic nature makes them difficult to purify and crystallize, traditional hurdles for techniques like X-ray crystallography.
Cryo-electron microscopy (Cryo-EM) has emerged as a transformative technology in structural biology, offering the ability to resolve the high-resolution 3D structures of biological macromolecules, including challenging targets like membrane proteins. Unlike crystallization-based methods, Cryo-EM allows samples to be preserved in a near-native hydrated state, overcoming some of the difficulties associated with membrane protein sample preparation. Shuimu BioSciences, founded in 2017 and stemming from Tsinghua University's structural biology heritage, is noted as the first commercial platform in Asia offering Cryo-EM structure determination services. They specialize in determining structures for a range of biomolecules, including membrane proteins like GPCRs, ion channels, and transporters.
The Challenges in Cryo-EM of Membrane Proteins
While Cryo-EM offers distinct advantages, determining the structure of membrane proteins using techniques like Single Particle Analysis (SPA) still presents unique challenges. These difficulties are often related to the inherent properties of membrane proteins and the requirements of the Cryo-EM workflow.
Sources indicate several challenges for Cryo-EM SPA in general, many of which are particularly relevant to membrane proteins:
- Small protein molecular weight: Some membrane protein targets, or domains used for structural study, might be small, making them harder to image and align. Shuimu has successfully elucidated protein structures as small as 51 kDa using Cryo-EM.
- Low concentration: Obtaining high concentrations of purified, stable membrane proteins can be difficult. Low sample concentration poses a challenge for data acquisition in Cryo-EM.
- High background noise: Biological samples are often imaged against a background of ice and grid material, contributing noise that can obscure small or low-contrast particles. Membrane proteins embedded in detergent micelles or lipid nanodiscs can be particularly challenging in this regard.
- Air-water interface disruption: Proteins can be damaged or preferentially orient at the air-water interface during sample freezing, leading to poor data quality and biased particle distributions. This is a common issue for many proteins, and membrane proteins are not immune.
- Preferential orientation: Particles often stick to the grid surface in a limited number of orientations, preventing the collection of a complete set of 2D views needed for accurate 3D reconstruction. This is a major bottleneck in Cryo-EM and significantly impacts data resolution and the ability to resolve anisotropic particles, which can include many membrane protein complexes.
Beyond these general Cryo-EM challenges, sources highlight the specific difficulties in preparing membrane proteins. Shuimu recognizes the need for a complete protein expression and purification platform to minimize variability from sample transport and standardize the entire pipeline, successfully addressing challenges posed by difficult-to-express proteins. They specifically mention having extensive experience in the production and purification method design of membrane proteins, including GPCRs, ion channels, and transport proteins. This underscores that the challenges for membrane protein Cryo-EM begin long before the sample goes into the microscope.
Breakthroughs and Solutions Offered by Cryo-EM
Despite these challenges, Cryo-EM has enabled significant breakthroughs in understanding membrane proteins. The ability to image samples in a near-native state and the continuous advancements in hardware and software have unlocked the structures of previously intractable targets. Shuimu BioSciences has been at the forefront, contributing to these breakthroughs through their services and proprietary technologies.
1. Addressing Sample Preparation Challenges: Shuimu provides comprehensive protein preparation and analysis services. This is crucial for membrane proteins, which require specialized techniques for expression and purification. Their platform features molecular cloning, protein expression using various systems (E. coli, mammalian cells, insect cells, cell-free), purification processes like affinity chromatography, ion-exchange chromatography, gel filtration, and RP-HPLC, and characterization using methods like SDS-PAGE, Western blot, mass spectrometry, and SPR/BLI/ELISA. By enhancing these upstream capabilities, Shuimu aims to provide "One-Stop" solutions from gene sequences to high-resolution 3D structures. They emphasize their extensive experience in the production and purification of membrane proteins like GPCRs, ion channels, and transporters. Shuimu also maintains an Off-the-Shelf Protein List which includes numerous membrane proteins like GPCRs, ion channels, and transporters, making samples of important drug targets readily available.
2. Overcoming Imaging Difficulties with Advanced Grids: To combat specific issues like preferential orientation, low concentration thresholds, high background noise, and air-water interface damage, Shuimu has developed a series of graphene support grids called GraFuture™. These grids, including Graphene Oxide (GO) and Reduced Graphene Oxide (RGO) versions, provide a potential solution to these bottlenecks. Their use in Cryo-EM is cited in research publications, suggesting their effectiveness in improving sample distribution and orientation.
3. Leveraging Advanced Instrumentation and Platform Scale: Shuimu operates one of the world's largest commercial Cryo-EM platforms, with 300 kV instruments available for 24-hour data acquisition (2 in Beijing, 6 in Hangzhou). This extensive hardware configuration, featuring advanced detectors, energy filters, spherical aberration correctors, and phase plates, is designed to ensure high imaging quality and facilitate rapid data acquisition. The scale allows for faster turnaround times and the ability to handle larger projects or more challenging samples that require extensive data collection. Regular maintenance ensures optimal equipment performance and stable service.
4. Driving Efficiency and Accuracy with AI: Data processing and 3D reconstruction are critical steps in Cryo-EM SPA. Shuimu utilizes proprietary AI algorithms and software suites, such as the SMART software suite for general Cryo-EM data analysis and NanoSMART for nanoparticle characterization (relevant for things like LNPs used to deliver molecules). These AI tools are designed to streamline the data analysis process, reduce machine runtime and required data volume, and enhance the overall efficiency and accuracy of structure determination. For MicroED, Shuimu developed eTasED software to integrate MicroED technology into conventional Cryo-EM systems, enhancing research efficiency and accuracy. While specifically mentioned for nanoparticles, the capabilities of AI in automatically identifying features, enhancing low-quality images, and performing accurate recognition and segmentation are beneficial for analyzing complex Cryo-EM datasets, including those from membrane proteins.
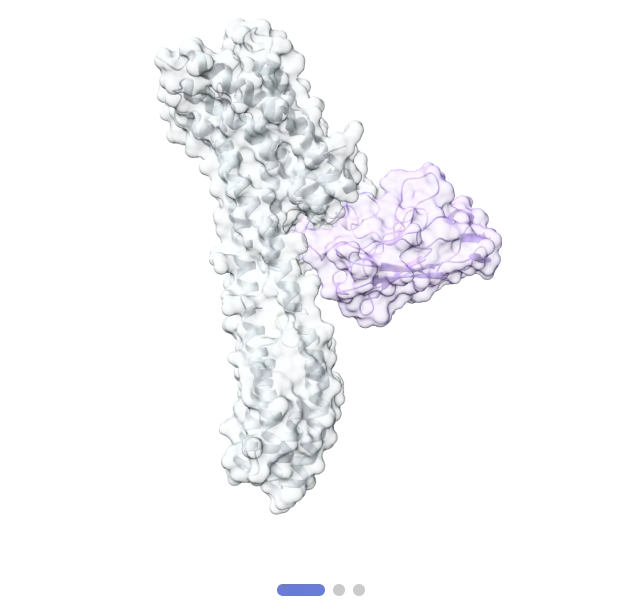
5. Achieving High Resolution and Tackling Complex Structures: Shuimu's platform has demonstrated impressive resolution capabilities, achieving a best resolution of 1.8Å and even a groundbreaking resolution of 1.4Å in some cases. They have successfully resolved structures of proteins as small as 51 kDa. Their extensive experience includes over 400 completed Cryo-EM projects and more than 150 resolved structures. Their case studies and publications showcase numerous high-resolution structures of membrane proteins like GPCRs, ion channels, and transporters, as well as their complexes with ligands or antibodies. Examples include the human GluN1-GluN2A subtype NMDA receptor bound with small molecules, the human histamine H1 receptor/Gq complex, structures revealing gating modulation of human Nav1.7, ligand recognition and activation mechanisms of chemokine receptors CCR2 and CCR3, insights into galanin receptor signaling, and structures of human polyamine transporter ATP13A2, multidrug transporter MRP4, and calcium pump SPCA1. These resolved structures represent significant cryo em breakthroughs for membrane proteins, providing atomic-level details crucial for understanding function and guiding drug design.
Applications in Drug Discovery and Antibody Development
Structural information obtained via Cryo-EM, particularly for key targets like membrane proteins, is invaluable for drug discovery and development.
- Understanding Mechanism of Action: Cryo-EM reveals how ligands, including potential drug candidates, bind to membrane proteins like GPCRs, shedding light on receptor activation and signal transduction mechanisms. This detailed understanding is critical for developing antibodies or small molecule drugs targeting these proteins. Shuimu offers SPR and BLI services to study molecular interactions, complementing their structural services.
- Antibody Drug Development: Many antibody drug targets are membrane proteins. Cryo-EM plays a crucial role by resolving the high-resolution 3D structures of antibody–antigen interactions. This helps researchers understand recognition mechanisms and binding sites, which is key for designing more effective antibody drugs. Cryo-EM is used to study how antibody drugs work, for instance, how they bind to targets and modulate signaling pathways. Structural analysis aids in identifying potential improvements in existing antibody drugs and supports the design of antibodies with higher affinity and specificity. Shuimu offers antibody discovery services, including screening, production, purification, and characterization, utilizing methods like ELISA, SPR, and BLI to validate binding activity and affinity. Their expertise in resolving structures of antigen-antibody complexes is evidenced by case studies and publications.
- Accelerating Development: With its high resolution and rapid data acquisition capabilities, Cryo-EM enables quick and detailed structural analysis, accelerating the optimization of drug designs and overall drug development processes.
Why Choose Shuimu BioSciences for Cryo-EM of Membrane Proteins?
Shuimu BioSciences positions itself as a leader in Cryo-EM services, offering several key advantages for tackling challenging targets like membrane proteins:
- One-Stop Solutions: Providing an integrated workflow from protein expression and purification to Cryo-EM structure determination and analysis. This minimizes sample variability and standardizes the entire pipeline, particularly beneficial for complex membrane protein projects.
- Cutting-Edge Equipment: Operating the world's largest commercial Cryo-EM platform with advanced 300 kV instruments and supporting apparatus.
- Elite Scientist Team: Composed of PhD-level experts specializing in structural biology, protein science, and computational biology with extensive experience in Cryo-EM and MicroED. Their experience specifically includes over 200 Cryo-EM projects spanning various targets, including membrane proteins and antigen-antibody complexes.
- Proprietary Technologies: Utilizing AI-driven platforms like SMART and NanoSMART, and graphene grids like GraFuture™, to enhance efficiency, accuracy, and overcome specific technical challenges.
- Extensive Experience with Membrane Proteins: Acknowledged expertise in producing and purifying membrane proteins and successfully resolving their structures, as demonstrated by their off-the-shelf list and published case studies.
- High Resolution: Uncompromising pursuit of resolution, achieving structures down to 1.8Å and even 1.4Å.
In summary, Cryo-EM has brought about significant breakthroughs in determining the structures of membrane proteins, overcoming many limitations of previous techniques. By addressing challenges in sample preparation, imaging, and data processing through integrated services, advanced technologies like graphene grids and AI, and extensive expertise, platforms like Shuimu BioSciences are accelerating the pace of structural biology and its application in drug discovery.
For more information on how Shuimu BioSciences can support your research projects involving membrane proteins and other challenging targets using their state-of-the-art cryo em services and breakthroughs, please visit https://shuimubio.com/.









Sign in to leave a comment.